Abstract
Background Major limitations to CD19-targeted chimeric antigen receptor T (CART19) cell therapy include suboptimal antitumor activity and development of life-threatening toxicities such as cytokine release syndrome (CRS) and neurotoxicity. CART-associated toxicities are directly related to CART activation and proliferation in vivo. Emerging data suggest that CART over-activation contributes to therapeutic failure.
Objective We hypothesized that modulation of CART activation reduces the risk of CRS while enhancing CART efficacy. Specifically, we hypothesized that using the clinically-approved CD19 monoclonal antibody tafasitamab prior to CART19 therapy can reduce tumor pyroptosis (inflammatory lytic programmed cell death), modulate CART activation, and enhance CART therapeutic index.
Results First, we tested the optimal sequencing of tafasitamab and CART19 in preclinical models. We used a 4-1BB costimulated CART19 construct, similar to FDA-approved tisagenlecleucel. Our data indicate that sequential use of tafasitamab followed by CART19 ameliorated overactivation, prevented apoptosis, and improved antitumor activity of CART19 in xenograft models (Sakemura et al, ASH 2021). Given the reduction of CART19 apoptosis after treatment with tafasitamab, we aimed to determine the effect of tafasitamab on tumor pyroptosis. Here, we measured the level of high mobility group box-1 (HMGB-1) in CD19+ tumor cells after co-culture with CART19 and tafasitamab. CD19 modulation with tafasitamab resulted in a significant reduction of HMGB-1, which indicates reduced tumor pyroptosis.
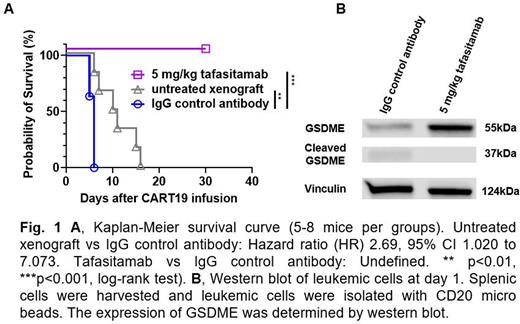
Given these in vitro results, we aimed to study how CD19 occupancy with tafasitamab prior to CART19 infusion impacts tumor pyroptosis in vivo. We utilized a mouse model that mimics CART toxicities. Immunocompromised NSG mice were inoculated via tail vein with 3-5x106 leukemic cells derived from patients with relapsed/refractory acute lymphoblastic leukemia (Sterner et al. Blood 2019, Sakemura et al. Cancer Immunol Res. 2021). Three weeks later, tumor burden was assessed by peripheral blood sampling. Leukemic cells were defined as murine CD45- and human CD45+CD19+CD20+ via flow cytometry, and volumetric absolute number was calculated. Mice were then randomized according to tumor burden to receive IgG control antibody one week before CART19 infusion [Group 1 (G1)], 5 mg/kg/day of tafasitamab one week before CART19 infusion (G2) or no treatment (G3). CRS was determined by weight loss, development of motor weakness, poor body posture, and elevated serum cytokine levels (e.g., IL-6, MCP-1, sCD40L, and GM-CSF). Prior to CART19 infusion, absolute number of CD19 molecules on tumor cells was quantified with antigen-binding capacity beads. G2 showed significant reduction of unbound/detectable CD19 molecules on the leukemic cell surface compared to G1 and G3. On day 0, 3x106 luciferase+CART19 were injected into G1 and G2 via tail vein. Mice were monitored with weekly bioluminescent imaging to track and quantify CART19 expansion and daily weighing and physical assessment to detect toxicity. G1 demonstrated significant weight reduction, motor weakness, and rapid CART19 expansion compared to G2. G1 reached an endpoint within 5 days of CART19 administration due to CRS, while G2 had delayed weight reduction and CART19 expansion. However, all mice from G2 remained healthy and did not display signs of CRS, including motor weakness and hunched posture. On day 5, CRS-related serum cytokines/chemokines were significantly elevated in G1 compared to G2. Overall survival was significantly longer in G2 (Fig 1A). Spleens were harvested, weighed, and assessed with flow cytometry. Spleens from G1 and G3 were significantly enlarged and were mainly composed of CD20+ leukemic cells compared to G2 spleens, which were smaller and infiltrated with CD3+ CART cells but not leukemic cells. Satellite mice were euthanized on day 1 and spleens were harvested. Western blot analysis revealed that mice from G1 showed significantly lower expression of apoptosis-inducing protein Gasdermin E (GSDME) and higher expression of cleaved GSDME on leukemic cells compared with G2, indicating reduced tumor pyroptosis in G2 (Fig 1B).
Conclusions In summary, CD19 binding with tafasitamab improved leukemic disease control and decreased the incidence and severity of CRS after CART19 administration in vivo, in part through the reduction of tumor pyroptosis.
Disclosures
Sakemura:Humanigen: Patents & Royalties. Cox:Humanigen: Patents & Royalties. Ilieva-Babinsky:Morphosys: Current Employment. Augsberger:Morphosys: Current Employment. Patra-Kneuer:Morphosys: Current Employment, Patents & Royalties. Heitmüller:Morphosys: Current Employment. Steidl:Morphosys: Current Employment. Parikh:Phamacyclics: Consultancy, Research Funding; Janssen: Research Funding; AstraZeneca: Consultancy, Research Funding; Merck: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; Ascentage Pharma: Research Funding; Genetech: Consultancy; GlaxoSmithKline: Consultancy; Adaptive Biotehcnologies: Consultancy; TG Therapeutics: Research Funding. Ding:Merck: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; AstraZeneca: Research Funding; BeiGene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Research Funding; Octapharma: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; DTRM: Research Funding; MEI pharama: Honoraria, Membership on an entity's Board of Directors or advisory committees; Alexion: Honoraria, Membership on an entity's Board of Directors or advisory committees. Kay:Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees, Other: Data Safety Monitoring Committee; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Cytomx Therapy: Membership on an entity's Board of Directors or advisory committees, Other: Data Safety Monitoring Committee; Beigene: Membership on an entity's Board of Directors or advisory committees; Behring: Membership on an entity's Board of Directors or advisory committees; Oncotracker: Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Morpho-sys: Other: Data Safety Monitoring Committee; Rigel: Other: Data Safety Monitoring Committee; Celgene: Other: Data Safety Monitoring Committee, Research Funding; Targeted Oncology: Membership on an entity's Board of Directors or advisory committees; Dren Bio: Other: Data Safety Monitoring Committee; BMS: Other: Data Safety Monitoring Committee, Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees, Other: Data Safety Monitoring Committee; AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Research Funding; MEI Pharma: Research Funding; Sunesis: Research Funding; TG Therapeutics: Research Funding; Tolero Pharmaceuticals: Research Funding. Nowakowski:Bantam Pharmaceutical: Consultancy; Blueprint Medicines Corporation: Consultancy; Celgene Corporation/Bristol Myers Squibb: Consultancy, Research Funding; Curis, Inc.: Consultancy; Daiichi Sankyo Inc: Consultancy; F. Hoffmann-La Roche Ltd: Consultancy, Research Funding; Genentech, Inc: Consultancy, Research Funding; Incyte: Consultancy; Karyopharm: Consultancy; Kite Pharma Inc.: Consultancy; Kymera Therapeutics: Consultancy; MorphoSys US Inc: Consultancy; NanoString: Research Funding; Ryvu Therapeutics: Consultancy; Selvita: Consultancy; TG Therapeutics: Consultancy; Zai Lab: Consultancy. Kenderian:Tolero: Research Funding; Viracta/Sunesis: Research Funding; Life Engine: Current holder of stock options in a privately-held company; Morphosys: Research Funding; LEAH Labs: Current holder of stock options in a privately-held company, Research Funding; MustangBio: Patents & Royalties; Novartis: Consultancy, Patents & Royalties: CART cell therapy , Research Funding, Speakers Bureau; Kite/Gilead: Consultancy, Research Funding, Speakers Bureau; Juno/BMS: Consultancy, Research Funding, Speakers Bureau; Lentigen: Research Funding; Humanigen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties: CART cell therapy , Research Funding, Speakers Bureau; Mettaforge: Patents & Royalties.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal